Development of the atomic theory
|
A scientist named Bohr proposed that electrons are located in levels at certain distances from the nucleus. The study was later revised and the electron cloud model was developed. The electron cloud model above represents the current atomic theory. An electron cloud is a region around the nucleus of an atom where electrons are likely to be found. Atoms are tiny, but scanning tunneling electron microscopes are used to display images of them.
|
Atoms...
Scientists have dome experiments that have revealed important clues about the structure of atoms. Atoms are the smallest unit of an element that maintains the properties of that element. Imagine cutting something into half billions of times. You will eventually get something that cannot be cut further. That is called an atom. Dalton's Atomic Theory 1) All matter is made of atoms. Atoms are indivisible and indestructible. 2) All atoms of a given element are identical in mass and properties 3) Compounds are formed by a combination of two or more different kinds of atoms. 4) A chemical reaction is a rearrangement of atoms. J.J. Thomson's discovery of the electron in 1897 showed us that the atom can be split into even smaller parts. His discovery was the first step towards a detailed model of the atom. Later, Rutherford made an experiment where he shot alpha rays through gold foil and he hypothesized that the alpha rays particles would pass through the gold foil with little or no deflection. However when he did the experiment he saw that although most of the alpha rays did pass right through, some of the particles were deflected greatly. This showed that the atom is made up of mainly empty space except for the nucleus in the middle which has the most mass in the atom. Ruth ford later Revised his plan in 1911 and found the nucleus. he reasoned that the positive charged particles were pushed away because of the positivity in the nucleus. |
the atom
Key Concept: An atom's properties are determined by the particles it is made of: protons, neutrons, and electrons.
Vocabulary
proton: A subatomic particle that has a positive charge and that is located in the nucleus of an atom
atomic mass unit: a unit of mass that describes the mass of an atom or molecule
neutron: a subatomic particle that has no charge and that is located in the nucleus of an atom
atomic number: the number of protons in the nucleus of an atom and this number is the same for all atoms of an element
isotope: an atom that has the same number of protons as other atoms of the same element do, but has a different number of neutrons, thus, changing the atomic mass
mass number: the sum of the numbers of protons and neutrons in the nucleus of an atom
atomic mass: the mass of an atom expressed in atomic mass units
Summary
The particles that make up an atom are protons, neutrons, and electrons. These three particles are called subatomic particles because they are much smaller than an atom.
PROTONS:
Even though electrons are much smaller, their charges are equal, so they cancel out.
ION➪an atom with a charge (from either gaining or losing electrons)
WHY NEUTRONS? Neutrons keep the protons from repelling or moving apart from each other, because positive charges repel from one another.
THE GENERIC ATOM
A generic atom has an equal number of protons and electrons, therefore they have a neutral charge. However, the number of neutrons does not have to equal the number of protons. In an element, the number of protons is the atomic number of that element. All atoms of the same element have the same atomic number, but not always the same atomic mass. ISOTOPES are atoms that have the same number of protons but have a different number of neutrons, and therefore a different atomic mass. Isotopes are always the same element, but they all differ slightly from one another. They can have different properties.
Vocabulary
proton: A subatomic particle that has a positive charge and that is located in the nucleus of an atom
atomic mass unit: a unit of mass that describes the mass of an atom or molecule
neutron: a subatomic particle that has no charge and that is located in the nucleus of an atom
atomic number: the number of protons in the nucleus of an atom and this number is the same for all atoms of an element
isotope: an atom that has the same number of protons as other atoms of the same element do, but has a different number of neutrons, thus, changing the atomic mass
mass number: the sum of the numbers of protons and neutrons in the nucleus of an atom
atomic mass: the mass of an atom expressed in atomic mass units
Summary
The particles that make up an atom are protons, neutrons, and electrons. These three particles are called subatomic particles because they are much smaller than an atom.
PROTONS:
- positive charge
- in the nucleus
- the mass is 1.7*10^-24 or 1 atomic mass unit (amu)
- neutral charge
- in the nucleus
- ≈1 amu, but neutrons have a greater mass than protons
- negative charge
- outside of nucleus in electron clouds
- ≈0 amu 1800 electron=1 proton
Even though electrons are much smaller, their charges are equal, so they cancel out.
- 1 proton charge= 1 electron charge
ION➪an atom with a charge (from either gaining or losing electrons)
- if the atom gains electrons, then it now has a negative charge
- if the atom loses electrons, then it now has a positive charge.
WHY NEUTRONS? Neutrons keep the protons from repelling or moving apart from each other, because positive charges repel from one another.
THE GENERIC ATOM
A generic atom has an equal number of protons and electrons, therefore they have a neutral charge. However, the number of neutrons does not have to equal the number of protons. In an element, the number of protons is the atomic number of that element. All atoms of the same element have the same atomic number, but not always the same atomic mass. ISOTOPES are atoms that have the same number of protons but have a different number of neutrons, and therefore a different atomic mass. Isotopes are always the same element, but they all differ slightly from one another. They can have different properties.
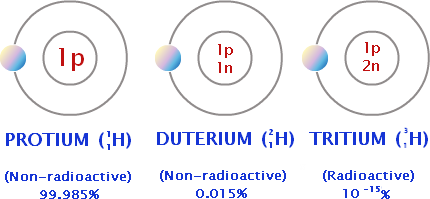
This is a picture of hydrogen atoms. They are all isotopes of the same element. They all have different properties ans a different atomic mass. Tritium is radioactive unlike protium and duterium. If an atom is radioactive, it is an unstable atom. This means that the nucleus of this atom will change over time and spontaneously fall apart, giving off small particles and energy. Each element has a limited number of isotopes. Isotopes can share chemical and physical properties. For example, an oxygen atom with isotopes of 8,9, and 10 neutrons are all colorless, odorless, and are gas at room temperature. We can also identify each isotope of an element by its mass number. Electrons are not included in the mass number because electrons are very tiny and their mass is almost nothing.
Naming Isotopes To identify a specific isotope of an element, write the name of the element followed by a hyphen and the mass number of the isotope.
ex: hydrogen; 1 proton, 1 neutron= Hydrogen-2
ISOTOPES AND ATOMIC MASS
Atomic mass or atomic weight is the average mass of atoms of an element, calculated using the relative abundance of isotopes in a naturally-occurring element. The weighted average is the percentages of each isotope that are present.
Ex: Copper: 69% copper-63 and 31% copper-65
Atomic Mass=63.6 amu
Forces in Atoms
The things that hold atoms together are gravitational force, electromagnetic force, strong force, and weak force. These work together to give an atom its structure and properties.
ex: hydrogen; 1 proton, 1 neutron= Hydrogen-2
ISOTOPES AND ATOMIC MASS
Atomic mass or atomic weight is the average mass of atoms of an element, calculated using the relative abundance of isotopes in a naturally-occurring element. The weighted average is the percentages of each isotope that are present.
Ex: Copper: 69% copper-63 and 31% copper-65
Atomic Mass=63.6 amu
Forces in Atoms
The things that hold atoms together are gravitational force, electromagnetic force, strong force, and weak force. These work together to give an atom its structure and properties.
elementsAn element is made up of only one type of atom and can be classified by a unique set of properties.
|
compoundsA compound is formed by chemically combining two or more elements, usually with different properties.
|
mixturesA mixture is a combination of two or more substances that are not chemically combined.
|